Engineered Bacteria to Fight Cancer Tumors From the Inside Out
Scientists at the University of Waterloo have designed bacteria that invade oxygen-starved tumor cores and consume cancer from within, in what could become a major new treatment approach
Feb 27, 2026
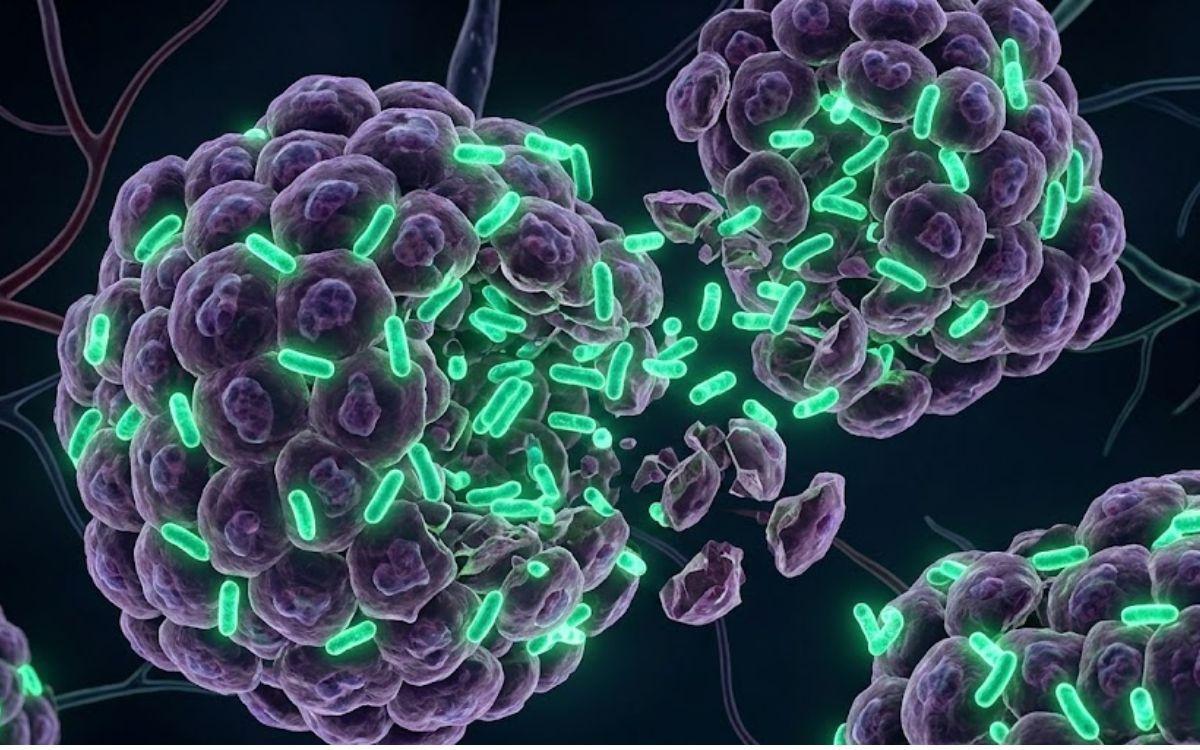
Cancer treatment has come a long way from chemotherapy and radiation. Now, researchers at the University of Waterloo in Canada are working on something that sounds like science fiction but is grounded in careful biology: bacteria engineered to invade tumors and eat them from the inside out.
The research, published in ACS Synthetic Biology, targets a structural weakness at the heart of solid tumors. Most of them have an oxygen-starved core made up of dead and dying cells. That dead zone is exactly what makes this approach possible.
How Engineered Bacteria Target Tumor Cores
According to ScienceDaily, the bacterium at the center of this research is Clostridium sporogenes, a microbe commonly found in soil that can only survive in environments with no oxygen at all. When its spores are introduced into the body, they naturally migrate toward and thrive inside the airless cores of solid tumors.
"Bacteria spores enter the tumor, finding an environment where there are lots of nutrients and no oxygen, which this organism prefers, and so it starts eating those nutrients and growing in size," said Dr. Marc Aucoin, a chemical engineering professor at Waterloo. "The bacterium is essentially ridding the body of the tumor."
Left alone, though, the bacteria run into a problem. As they multiply and expand outward toward the tumor's edges, they encounter small amounts of oxygen and begin to die off before the job is finished. The cancer at the outer regions of the tumor survives.
To fix this, the research team inserted a gene from a related bacterium that has higher oxygen tolerance. This modification allows the engineered microbes to survive longer as they push toward the outer edges of the tumor.
Quorum Sensing: The Safety Switch
Turning on that oxygen-tolerance feature too early would be dangerous. If the bacteria activated it before entering the tumor, they could potentially survive and grow in oxygen-rich environments like the bloodstream. The researchers needed a built-in safety mechanism.
The solution they found is called quorum sensing, a natural communication process that bacteria already use. As bacterial numbers grow, each microbe releases a chemical signal. When enough bacteria accumulate in one place, the combined signal crosses a threshold and triggers a genetic response.
The team programmed this natural system to act as a switch. Only after enough bacteria have gathered inside the tumor, confirming they are in the right location, does the signal grow strong enough to turn on the oxygen-tolerance gene. This means the bacteria essentially know where they are before activating their survival mechanism.
According to ScienceDaily, Dr. Brian Ingalls, a professor of applied mathematics at Waterloo, described the approach this way:
"Using synthetic biology, we built something like an electrical circuit, but instead of wires we used pieces of DNA. Each piece has its job. When assembled correctly, they form a system that works in a predictable way."
From Lab to Pre-Clinical Trials
In an earlier study, the team confirmed that Clostridium sporogenes could be genetically altered to better withstand oxygen. In a follow-up experiment, they tested their quorum sensing design by programming bacteria to produce a green fluorescent protein, allowing them to verify the system activated exactly when intended.
The next step is to combine both the oxygen-tolerance gene and the quorum-sensing control system into a single engineered bacterium and test it against tumors in pre-clinical trials.
The research began as PhD work by Bahram Zargar, supervised by Dr. Ingalls and Dr. Pu Chen. Dr. Sara Sadr, a former Waterloo doctoral student, played a leading role in advancing the project. The team is now collaborating with CREM Co Labs, a Toronto-based microbiology company co-founded by Dr. Zargar.
The approach is not the first attempt to use bacteria against cancer, but the precision engineering here sets it apart. Previous bacterial cancer therapies struggled with the same oxygen-barrier problem. The quorum-sensing control system is what makes this version potentially safer and more targeted than earlier attempts.
Researchers are cautious about timelines, as pre-clinical trials still lie ahead before any human application. But the science is sound, peer-reviewed, and addresses a real limitation in treating solid tumors, which make up roughly 90% of all cancers. Conventional treatments like chemotherapy often cannot penetrate the necrotic core of these tumors effectively, which is precisely the zone these bacteria are designed to occupy.
Stay tuned for more updates and news!




